THE FUTURE OF PRECISION MEDICINE
Precision medicine’s future relies on medical research using data in new and innovative ways. By leveraging a patient’s unique biological response to disease or injury, we are developing targeted clinical decision support tools (CDSTs) that will enhance surgical decision making, thereby reducing the cycle of care (e.g. trips to the operating room, time in the intensive care unit or general ward), lowering resource utilization, and improving clinical outcomes.
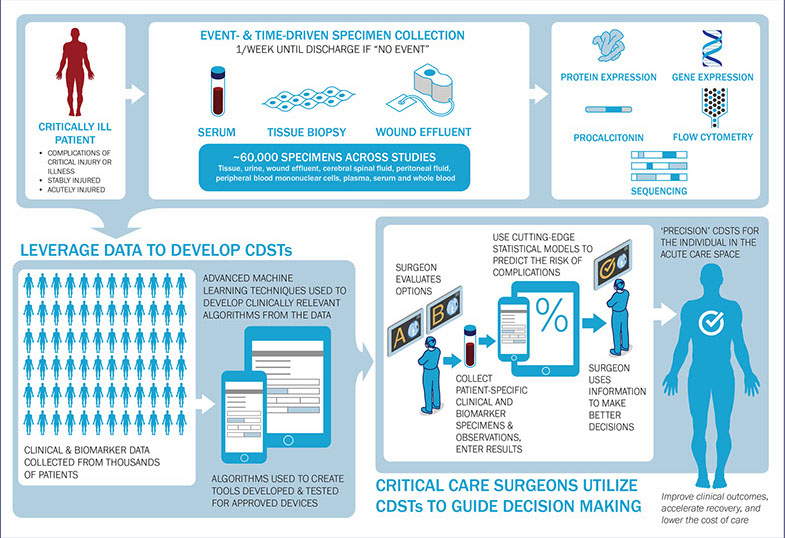
We leverage a growing biobank of approximately 70,000 specimens collected longitudinally from critically ill patients to develop a robust databank of 40 million data elements. In turn, these elements are powering the development of algorithms able to predict complications for conditions typically associated with a high risk of morbidities or mortality (e.g. blood clots, acute respiratory distress syndrome, acute kidney injury, sepsis and pneumonia).
Our team has enrolled 320 service members injured during Operation Enduring Freedom (OEF), Operation Iraqi Freedom (OIF) and 1,500 critically ill civilian and military patients in four clinical trials. In both civilian and military health systems, rates of surgical complications for traumatic extremity injuries can reach 15 percent. Leveraging a patient’s individual biology and personalizing both diagnoses and therapeutic interventions, our CDSTs are expected to improve these clinical outcomes from 85 percent to 95 percent. Our CDSTs further assist readiness by either accelerating return to duty (abridged length-of-stay across the ICU, General Ward and the rehabilitation continuum of care) or curbing logistical burdens (e.g., reduced need for blood products and airlifts) during conflicts. We have already deployed three CDSTs that predict sepsis in medical and surgical Intensive Care Units (ICUs), the incidence of invasive fungal infection, and the need to activate a massive transfusion protocol.
Presently, there are 40 ongoing analytics projects and approximately 10 additional biomarker-based tools that are in various stages of development, of which two are linked to electronic health records. The first of a series of FDA clinical trials launched in fiscal year 2020. In the future, we intend to leverage our growing databank to develop a battery of predictive algorithms for conditions associated with high mortality and morbidity. These tools are expected to generate substantial annual cost savings in both civilian ($10 billion) and military ($111 million) health systems.
Figure 2. Integrative and Standardized Approach to Clinical & Biomarker Data Collection
NEW AND PROVEN METHODS MAXIMIZE OUTCOMES
Our model of providing individualized treatment based on underlying biology has the potential to transform medicine with data that improves patient outcomes — while reducing costs.
Wound Healing/ Local Complications
Extremity injuries (timing of closure, complications)
Open abdomen (timing of closure, complications)
Severe Traumatic Brain Injury (vasospasm, mortality)
Small Bowel Obstruction (risk prediction)
Systemic Complications
Venous Thromboembolism (risk prediction)
Acute Respiratory Distress Syndrome (risk prediction)
Acute Kidney Injury (risk prediction)
Heterotopic Ossification (risk prediction)
Infections / Infectious Complications
Bacteremia (risk prediction)
>Pneumonia (risk prediction)
Host-pathogen Interactions (mechanistic model)
Sepsis / Decompensation (risk prediction)
Appendicitis (risk prediction)
ONGOING STUDIES
These methods help maximize outcomes across any discipline requiring complex medical decision making, including surgery, critical care, emergency medicine, orthopedics, transplant and oncology.
Tissue and Data Acquisition Protocol (TDAP): A standardized method for collecting clinical data and biological specimens from properly identified patients and healthy volunteers.
Timing of Debridement and Closure (WounDX): A validated CDST is designed to guide the timing of wound closure in traumatic wounds as well as to establish criteria from both the number and frequency of wound debridement, on a wound specific basis.
Invasive Fungal Infections (IFI): Developing predictive models using SC2i’s technology to diagnose invasive fungal infections, devastating infections, early and institute treatment at an accelerated pace.
Massive Transfusion Protocol: A smart phone application created to allow the accurate prediction of massive transfusion based on a sophisticated statistical model created using admission variables readily available to the clinician at the bedside.
Damage Control Laparotomy – Open Abdomen: SC2i will conduct a prospective observational trial to define biomarkers as objective criteria that will be used to develop a CDST, for this purpose.
Traumatic Brain Injury (TBI): Future development of an accurate CDST to assist clinicians in understanding immunologic implications with a mechanistic focus directed towards clinically translatable therapeutic manipulations.
Decompensation (Decomp): The Physiologic Decompensation project will provide real-time analytics to create situation awareness to enhance care. The general intent of this project is to accelerate individualized treatments and provide rapid decision support based on the early detection of physiologic decompensation with the overall tripartite goal of better health, better care and lower costs for both military and civilian populations.